NK cell-based therapies
Research focus:
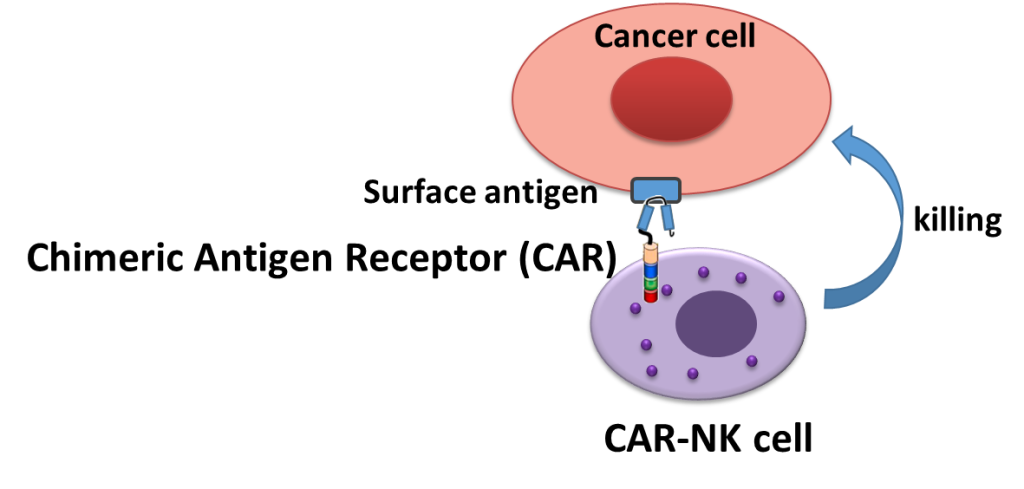
Cell therapy is a promising strategy for treating tumors where conventional therapies fail. Treatment with chimeric antigen receptor (CAR) T cells has been successful in selected hematological malignancies but until now by and large failed in solid tumors. NK cells are currently being developed as an alternative strategy and promise to be more effective in the treatment of solid tumors. NK cells are also more universally applicable without the risk of graft-versus-host disease (GvHD) and with favorable cytokine profile that protect from side effects after adoptive transfer. In recent years, our group and others have developed protocols for the clinical application of various NK cell sources. We have established NK-92 cells as clinically applicable cell line and proved clinical safety. We have further developed several CAR-retargeted NK-92 cell, where ERBB2-specific CAR-NK-92 cells are currently tested in clinical trial CAR2BRAIN (NCT03383978). We are further working on improvement of the CAR technology and NK cell-based therapies. We are also very interested in resistance mechanisms that lead to cancer cell escape from the NK cell immunosurveillance. Recently, we broaden our focus to non-cancer CAR-NK targets to treat various severe diseases.
Selected publications:
NK cells
Eitler J, Rackwitz W, Wotschel N, Gudipati V, Shankar NM, Sidorenkova A, Huppa JB, Ortiz-Montero P, Opitz C, Künzel SR, Michen S, Temme A, Loureiro LR, Feldmann A, Bachmann M, Boissel L, Klingemann H, Wels WS, Tonn T. 2024. CAR-mediated targeting of NK cells overcomes tumor immune escape caused by ICAM-1 downregulation. Journal for ImmunoTherapy of Cancer 12: e008155
Murali Shankar N, Ortiz-Montero P, Kurzyukova A, Rackwitz W, Künzel SR, Wels WS, Tonn T, Knopf F, Eitler J. 2023. Preclinical assessment of CAR-NK cell-mediated killing efficacy and pharmacokinetics in a rapid zebrafish xenograft model of metastatic breast cancer. Frontiers in Immunology 14
Silvestre RN, Eitler J, de Azevedo JTC, Tirapelle MC, Fantacini DMC, de Souza LEB, Swiech K, Covas DT, Calado RT, Montero PO, Malmegrim KCR, Figueiredo ML, Tonn T, Picanco-Castro V. 2023. Engineering NK-CAR.19 cells with the IL-15/IL-15Ralpha complex improved proliferation and anti-tumor effect in vivo. Frontiers in Immunology 14: 1226518
Steenblock C, Eitler J, Oikonomakos IT, Arriens M, Künzel SR, Tonn T, Bornstein SR. 2023. Application of chimeric antigen receptor-natural killer cells for the treatment of type 1 diabetes. Exploration of Endocrine and Metabolic Disease 1: 4-11
Wotschel N, Montero PO, Wels WS, Tonn T, Eitler J. 2022. Live-cell imaging for analysis of the NK cell immunological synapse. Methods in Cell Biology
Eitler, J., N. Wotschel, N. Miller, L. Boissel, H.G. Klingemann, W. Wels, and T. Tonn. 2021. Inability of granule polarization by NK cells defines tumor resistance and can be overcome by CAR or ADCC mediated targeting. J Immunother Cancer 9(1)
Mitwasi, N., A. Feldmann, C. Arndt, S. Koristka, N. Berndt, J. Jureczek, L.R. Loureiro, R. Bergmann, D. Mathe, N. Hegedus, T. Kovacs, C. Zhang, P. Oberoi, E. Jager, B. Seliger, C. Rossig, A. Temme, J. Eitler, T. Tonn, M. Schmitz, J.C. Hassel, D. Jager, W.S. Wels, and M. Bachmann. 2020. “UniCAR”-modified off-the-shelf NK-92 cells for targeting of GD2-expressing tumour cells. Scientific reports 10:2141.
Nowakowska, P., A. Romanski, N. Miller, M. Odendahl, H. Bonig, C. Zhang, E. Seifried, W.S. Wels, and T. Tonn. 2017. Clinical grade manufacturing of genetically modified, CAR-expressing NK-92 cells for the treatment of ErbB2-positive malignancies. Cancer immunology, immunotherapy : CII
Oelsner, S., M.E. Friede, C. Zhang, J. Wagner, S. Badura, P. Bader, E. Ullrich, O.G. Ottmann, H. Klingemann, T. Tonn, and W.S. Wels. 2017. Continuously expanding CAR NK-92 cells display selective cytotoxicity against B-cell leukemia and lymphoma. Cytotherapy 19:235-249.
Romanski, A., C. Uherek, G. Bug, E. Seifried, H. Klingemann, W.S. Wels, O.G. Ottmann, and T. Tonn. 2016. CD19-CAR engineered NK-92 cells are sufficient to overcome NK cell resistance in B-cell malignancies. Journal of cellular and molecular medicine 20:1287-1294.
Zhang, C., M.C. Burger, L. Jennewein, S. Genssler, K. Schonfeld, P. Zeiner, E. Hattingen, P.N. Harter, M. Mittelbronn, T. Tonn, J.P. Steinbach, and W.S. Wels. 2016. ErbB2/HER2-Specific NK Cells for Targeted Therapy of Glioblastoma. Journal of the National Cancer Institute 108:
Schonfeld, K., C. Sahm, C. Zhang, S. Naundorf, C. Brendel, M. Odendahl, P. Nowakowska, H. Bonig, U. Kohl, S. Kloess, S. Kohler, H. Holtgreve-Grez, A. Jauch, M. Schmidt, R. Schubert, K. Kuhlcke, E. Seifried, H.G. Klingemann, M.A. Rieger, T. Tonn, M. Grez, and W.S. Wels. 2015. Selective inhibition of tumor growth by clonal NK cells expressing an ErbB2/HER2-specific chimeric antigen receptor. Molecular therapy : the journal of the American Society of Gene Therapy 23:330-338.
Tonn, T., D. Schwabe, H.G. Klingemann, S. Becker, R. Esser, U. Koehl, M. Suttorp, E. Seifried, O.G. Ottmann, and G. Bug. 2013. Treatment of patients with advanced cancer with the natural killer cell line NK-92. Cytotherapy 15:1563-1570.
Esser, R., T. Muller, D. Stefes, S. Kloess, D. Seidel, S.D. Gillies, C. Aperlo-Iffland, J.S. Huston, C. Uherek, K. Schonfeld, T. Tonn, N. Huebener, H.N. Lode, U. Koehl, and W.S. Wels. 2012. NK cells engineered to express a GD2 -specific antigen receptor display built-in ADCC-like activity against tumour cells of neuroectodermal origin. Journal of cellular and molecular medicine 16:569-581.
Muller, T., C. Uherek, G. Maki, K.U. Chow, A. Schimpf, H.G. Klingemann, T. Tonn, and W.S. Wels. 2008. Expression of a CD20-specific chimeric antigen receptor enhances cytotoxic activity of NK cells and overcomes NK-resistance of lymphoma and leukemia cells. Cancer immunology, immunotherapy : CII 57:411-423.
Romanski, A., G. Bug, S. Becker, M. Kampfmann, E. Seifried, D. Hoelzer, O.G. Ottmann, and T. Tonn. 2005. Mechanisms of resistance to natural killer cell-mediated cytotoxicity in acute lymphoblastic leukemia. Experimental hematology 33:344-352.
Uherek, C., T. Tonn, B. Uherek, S. Becker, B. Schnierle, H.G. Klingemann, and W. Wels. 2002. Retargeting of natural killer-cell cytolytic activity to ErbB2-expressing cancer cells results in efficient and selective tumor cell destruction. Blood 100:1265-1273.
Dendritic cells and macrophages
Jacome-Galarza, C.E., G.I. Percin, J.T. Muller, E. Mass, T. Lazarov, J. Eitler, M. Rauner, V.K. Yadav, L. Crozet, M. Bohm, P.L. Loyher, G. Karsenty, C. Waskow, and F. Geissmann. 2019. Developmental origin, functional maintenance and genetic rescue of osteoclasts. Nature 568:541-545.
Percin, G.I.*, J. Eitler*, A. Kranz, J. Fu, J.W. Pollard, R. Naumann, and C. Waskow. 2018. CSF1R regulates the dendritic cell pool size in adult mice via embryo-derived tissue-resident macrophages. Nature communications 9:5279. (*equally contributed as first authors)